Novel Model of Antibody Mediated Rejection of Kidney Allografts in Rats, A
Department of Surgery, University of Wisconsin, Madison, WI
Department of Medicine, Division of Nephrology, University of Wisconsin, Madison, WI
Department of Pathology, University of Wisconsin, Madison, WI
Immunology, The Cleveland Clinic Foundation, Cleveland, OH
Meeting: 2013 American Transplant Congress
Abstract number: 355
We have developed a new experimental model of acute ABMR, in which complete MHC mismatched kidneys are transplanted from Brown Norway rats (RT1n) into Lewis (RT1l) recipients 3weeks after donor-specific blood transfusion (group G6). To reduce T cell alloreactivity, some sensitized recipients were treated with CsA post-transplant (G4).
| Group | Treatment | BUN | Scr | DSA (IgG2c) | C4d on circulating lymphocytes | CD3 | CD68 |
| G1 | – | 20.3 | 0.4 | 59.1 | 42.7 | – | +/- |
| G2 | TF | 19.3 | 0.4 | 245.67 | 840.33 | – | +/- |
| G3 | TX + CsA | 41.4 | 0.7 | 51.23 | 36.13 | – | +/- |
| G4 | TF + TX + CsA | 51.3 | 0.9 | 207 | 589 | + | ++ |
| G5 | TX | 219.4 | 3.5 | 152.23 | 213.75 | ++ | +++ |
| G6 | TF + TX | 135.8 | 2 | 403.33 | 610.67 | + | ++++ |
G6 had reduced kidney function, increased DSA (antidonor splenocyte IgG1, IgG2b and IgG2c), capillaritis, glomerulitis, and greater C4d staining in peritubular capilaries and on circulating lymphocytes consistent with ABMR.
| Group | C4d | ABMR Grade | ACR Grade | IL-2* | IL-4* | IL-10* | IFNgΓ* |
| G1 | – | – | 0 | 1 | 1 | 1 | 1 |
|---|---|---|---|---|---|---|---|
| G2 | – | – | 0 | 1.01 | 1.77 | 2.7 | -1.01 |
| G3 | minimal | – | 0 | 4.35 | 1.73 | 47 | 1.08 |
| G4 | Focal-Diffuse | II | IA-IIB | 9.72 | 3.88 | 15 | 25.3 |
| G5 | Diffuse | II | IA-IIA | 11.3 | 1.91 | 127 | 30.5 |
| G6 | Diffuse | II | Ia-III | 40.88 | 2.96 | 63 | 358 |
In addition, immunofluorescence studies demonstrated a greater CD68/CD3 ratio in kidneys with ABMR. Cytokine gene analyses demonstrated predominant Th1 (IFN-Γ, IL2) and Th2 (IL10) profiles in sensitized and non-sensitized recipients respectively. Immunoblot analyses showed a significant increase in IRF5, a transcription factor associated with M1 phenotype macrophages, as well as CRRY and CD55, complement regulatory proteins (CRPs) involved in the inhibition of C3 convertase.
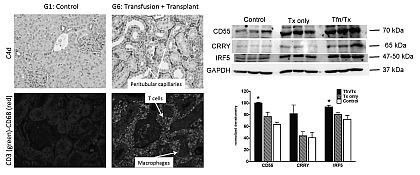
In conclusion, pretransplant blood transfusion was associated with ABMR, a Th1 cytokine profile, M1 macrophage phenotype and upregulation of CRPs. This animal model may facilitate studies to delineate the cellular and molecular pathways that regulate ABMR.
To cite this abstract in AMA style:
Huang G, Wilson N, Reese S, Jacobson L, III WBaldwin, Zhong W, Djamali A. Novel Model of Antibody Mediated Rejection of Kidney Allografts in Rats, A [abstract]. Am J Transplant. 2013; 13 (suppl 5). https://atcmeetingabstracts.com/abstract/novel-model-of-antibody-mediated-rejection-of-kidney-allografts-in-rats-a/. Accessed May 24, 2026.« Back to 2013 American Transplant Congress
