Monitoring Recipients of Public Health Service (PHS) Increased Risk Donors Using an Electronic Medical Record-Based System
1Yale School of Medicine, New Haven, CT, 2Yale New Haven Health, New Haven, CT
Meeting: 2020 American Transplant Congress
Abstract number: A-242
Keywords: Infection, Monitoring, Safety, Screening
Session Information
Session Name: Poster Session A: Quality Assurance Process Improvement & Regulatory Issues
Session Type: Poster Session
Date: Saturday, May 30, 2020
Session Time: 3:15pm-4:00pm
 Presentation Time: 3:30pm-4:00pm
Presentation Time: 3:30pm-4:00pm
Location: Virtual
*Purpose: In 2014, the Organ Procurement and Transplantation Network (OPTN) policy was updated, mandating transplant centers to implement a post-transplant screening protocol for transmissible diseases. The Public Health Service (PHS) Guideline recommends post-transplant screening of recipients of increased risk donor (IRD) organs for HIV, HBV, and HCV transmission. Screening was suggested at 1-3 months for HIV, HBV, HCV, and at 12 months for HBV post-transplant. Despite these recommendations, only 60% underwent post-transplant screening of recipients of PHS IRD in a multicenter survey. Barriers to implementation of screening included poor physician and patient follow-through.
*Methods: Since 2015, our center has tracked HIV, HBV and HCV screening of recipients of PHS IRD by an electronic medical record (EMR)-based system to improve patient safety and policy compliance. Recipients of IRD organs were consented as per policy, labeled in our EMR as PHS IRD, and automatically entered into an EMR-generated report. Clinicians and coordinators received a best practice alert (BPA) when the EMR was accessed during follow-up visits. The BPA served as a reminder for 1st screening (28 +14 days) and 2nd screening (1 year +21 days). The current study included patients who underwent organ transplantation between 1/1/2016 and 11/30/2018. We calculated the proportions of recipients who underwent 1st and 2nd screening and the time to 1st and 2nd screening.
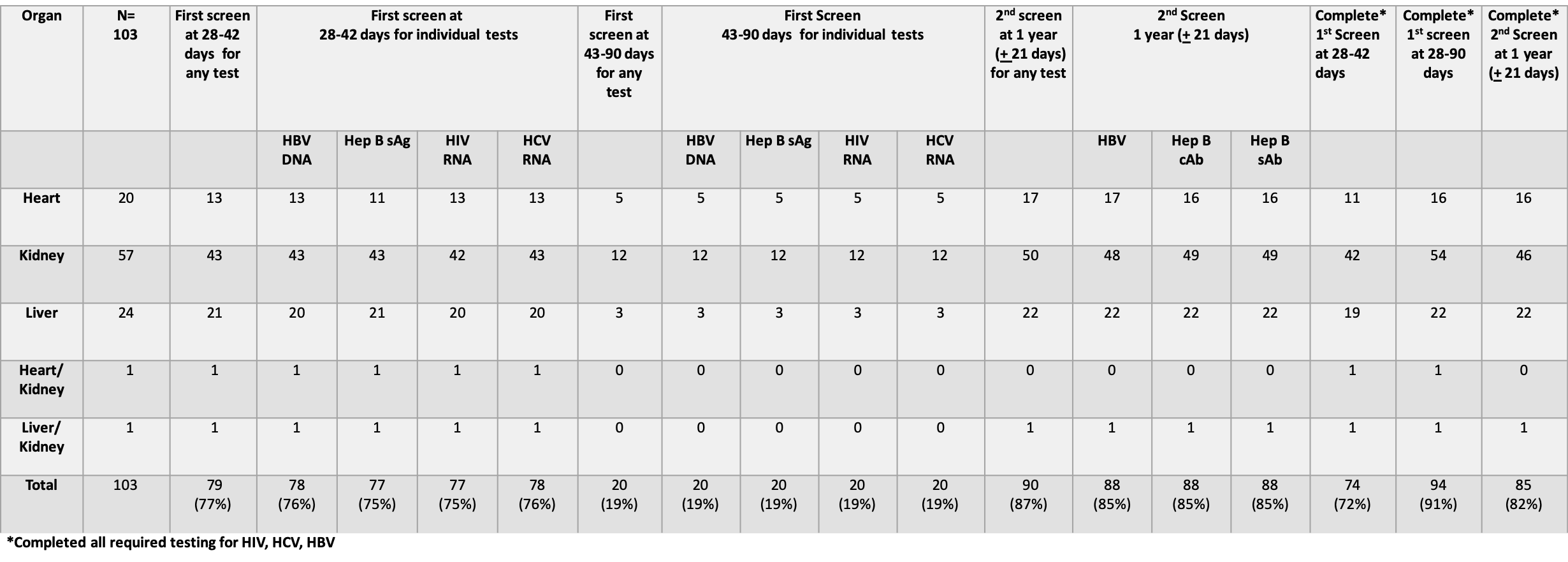
*Results: There were 103 recipients of IRD with a median follow-up of 859 days [345-1404 days]. All recipients underwent HIV, HBV, HCV screening post-transplant at some point. The median time to 1st screening was 37 days [18-126 days]. Seventy nine recipients (77%) underwent screening at 28-42 days, 20 (19%) at 42-90 days, and 4 (4%) at < 28 or > 90 days. The median time for 2nd screening was 355 days [324-460 days]. Ninety recipients (87%) underwent 2nd screening at 1 year [+ 21 days], while 9 (9%) were screened beyond the allotted time. Four recipients (4%) had no 2nd screening test; 3 of 4 had testing ordered but not completed due to patient compliance. No transmissions of HIV, HBV, and HCV infection occurred.
*Conclusions: Using an EMR-based system, our center achieved a high rate of screening post-transplantation; higher than in previously reported multicenter data. In light of proposed future universal recipients screening regardless of donor risk, EMR-based monitoring may improve compliance with scheduled testing and consequentially enhance patient safety.
To cite this abstract in AMA style:
Malinis M, Nuesse B, Carter S, Emre S, Formica R, Kulkarni S, Mulligan D, Schilsky M, Azar MM. Monitoring Recipients of Public Health Service (PHS) Increased Risk Donors Using an Electronic Medical Record-Based System [abstract]. Am J Transplant. 2020; 20 (suppl 3). https://atcmeetingabstracts.com/abstract/monitoring-recipients-of-public-health-service-phs-increased-risk-donors-using-an-electronic-medical-record-based-system/. Accessed May 31, 2026.« Back to 2020 American Transplant Congress