Managing the Costs of Routine Follow-up Care After Living Kidney Donation: A Survey of Contemporary Experience, Practice & Challenges
1Saint Louis University, Saint Louis, MO, 2Univ. Iowa, Iowa City, IA, 3Toyon Associates, Concord, CA, 4Univ. Wisconsin, Madison, WI, 5New York-Presbyterian Hosp., New York, NY, 6St. Barnabas, Livingston, NJ
Meeting: 2021 American Transplant Congress
Abstract number: 471
Keywords: Economics, Living donor, Public policy, Safety
Topic: Clinical Science » Kidney » Kidney Living Donor: Other
Session Information
Session Time: 7:30pm-8:30pm
 Presentation Time: 7:50pm-8:00pm
Presentation Time: 7:50pm-8:00pm
Location: Virtual
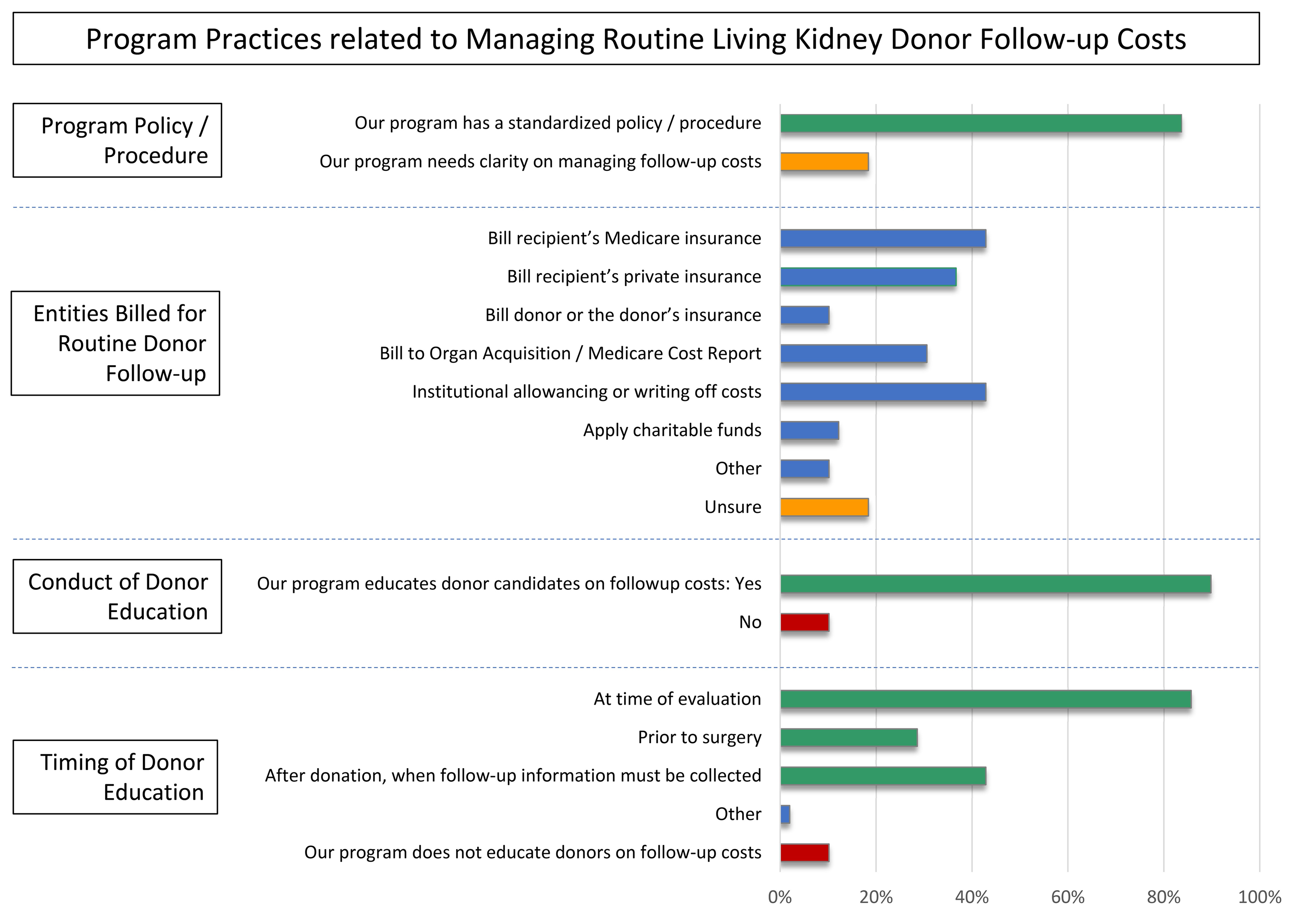
*Purpose: Organ Procurement and Transplantation Network (OPTN) Policy mandates that living kidney donor (LKD) programs collect and report clinical and laboratory follow-up information for living organ donors at 6 months, 1 year and 2 years postdonation. Handling follow-up costs can pose challenges for programs and donors. To help inform the community and guide discussions of effective strategies for managing follow-up costs, we surveyed US LKD programs on current experiences and practices.
*Methods: A brief 4-item survey instrument was designed by a multidisciplinary workgroup of professionals in transplant administration and LKD clinical care to assesses program policies, billing practices and donor candidate/donor education. We distributed the survey to staff at U.S. LKD transplant programs by email and posting to professional society list-servs in Fall 2020, using the Qualtrics survey platform in Fall 2020.
*Results: Among 49 programs that participated to date, 84% have a standardized policy and procedure on how to handle the costs of routine follow-up. Mechanisms for recovering or covering the costs of mandated routine postdonation follow-up at responding programs include: billing recipients’ Medicare (43%), billing recipients’ private insurance (37%), institutional allowancing (43%), billing to organ acquisition or Medicare Cost Report (MCR) (31%), billing donors/donors’ insurance (10%), and use of charitable funds (12%) (Figure). Although 90% of programs educate donor candidates on the costs and coverage of OPTN mandated follow-up, 10% do not. Provision of donor education most commonly occurs at the time of evaluation (89%), and 42% also educate at the time when follow-up must be conducted. 18% of programs are interested in more clarity on strategies for covering follow-up costs.
*Conclusions: Based on a pilot survey of U.S. LKD programs, a variety of approaches are used to cover routine postdonation follow-up, ranging from billing recipients’ or donors’ insurance, to institutional allowancing or use of charitable funds, to inclusion on the MCR. Notably, although Medicare disallowed including these expenses on the MCR or billing the recipients’ Medicare in 2016, these practices remain common, indicating a need for increased guidance. Up-to-date resources on handling the costs of routine donor follow-up will be useful for programs and living donors.
To cite this abstract in AMA style:
Lentine K, McNatt G, Howey R, Thomas C, Hays R, Lebron-Banks U, Ainapurapu S, Tietjen A. Managing the Costs of Routine Follow-up Care After Living Kidney Donation: A Survey of Contemporary Experience, Practice & Challenges [abstract]. Am J Transplant. 2021; 21 (suppl 3). https://atcmeetingabstracts.com/abstract/managing-the-costs-of-routine-follow-up-care-after-living-kidney-donation-a-survey-of-contemporary-experience-practice-challenges/. Accessed May 14, 2026.« Back to 2021 American Transplant Congress