Long Term Outcomes of Highly-HLA Sensitized (PRA>80%) (HS) Patients Receiving Desensitization with IVIG or IVIG + Rituximab: A Retrospective, Single-Center Analysis
Comprehensive Transplant Center, Cedars-Sinai Medical Center, Los Angeles, CA
Meeting: 2013 American Transplant Congress
Abstract number: A841
INTRODUCTION: Desensitization protocols (DES) for highly-HLA sensitized (PRA>80%) (HS) living donor and deceased donor recipients include low-dose IVIG + plasma exchange, high-dose (HD) IVIG and HD-IVIG + rituximab. Long-term data on efficacy and outcomes of each protocol is limited. Here, we undertook a retrospective analysis of two protocols (HD-IVIG v. HD-IVIG + rituximab). PATIENTS & METHODS: Between 3/02 and 10/09, we identified 30 HS patients who received DES with IVIG alone (IVIG 2gm/kg 2-4 doses given monthly). This was compared to 122 HS patients DES with IVIG +rituximab from 3/05-12/11 (IVIG 2gm/kg, weeks 1 & 4 + rituximab 1gm week 2). We analyzed effects of therapy on DSA and CMXs at transplant and ABMR. In addition, we assessed, graft & patient survival and attribution of graft loss. Induction therapy for the IVIG group was either Thymoglobulin® or Zenapax® while the IVIG + rituximab group received Campath-1H. All were maintained on Prograf® Cellcept® and prednisone. RESULTS: (see Table 1) Briefly, no differences in patient demographics were seen. Forty five percent patients in the IVIG group had previous transplants v. 63% in the IVIG + rituximab (p=0.09). However, T-cell FCMXs were significantly greater at pre-treatment and transplant in the IVIG + rituximab group. B-cell FCMXs were not different. DES with IVIG alone was associated with a significantly greater risk for ABMR (p=0.02) and graft loss from ABMR (p=0.0001). This is despite a similar DSA-RIS score at transplant and at ABMR. Two patients in the IVIG DES group lost grafts to BKAN.
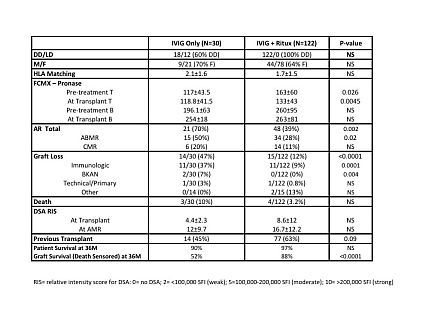
CONCLUSIONS: Although limited by time, DSA assessment technology and induction protocols, it is clear that DES with IVIG alone was associated with a significant risk for ABMR and graft loss from ABMR. Most patients developing ABMR in the IVIG alone group did so within 1M. The DSA scores at ABMR were similar, but the intensity of ABMR episodes were worse in the IVIG alone group (6 ABMR with TMA, 4 treated with eculizumab) compared to 1 eculizumab treated patient in the IVIG + rituximab group.
Jordan, S.: Grant/Research Support, Genentech, CSL Behring.
To cite this abstract in AMA style:
Vo A, Choi J, Kahwaji J, Peng A, Villicana R, Najjar R, Jordan S. Long Term Outcomes of Highly-HLA Sensitized (PRA>80%) (HS) Patients Receiving Desensitization with IVIG or IVIG + Rituximab: A Retrospective, Single-Center Analysis [abstract]. Am J Transplant. 2013; 13 (suppl 5). https://atcmeetingabstracts.com/abstract/long-term-outcomes-of-highly-hla-sensitized-pra80-hs-patients-receiving-desensitization-with-ivig-or-ivig-rituximab-a-retrospective-single-center-analysis/. Accessed May 21, 2026.« Back to 2013 American Transplant Congress
