It's Never too Early to Test: Frequent Anti-A2 Titer Monitoring in Blood Group B Patients is Necessary.
Medstar Georgetown Transplant Institute, Washington, DC
Meeting: 2017 American Transplant Congress
Abstract number: D305
Keywords: Allocation, Antibodies, Kidney transplantation
Session Information
Session Name: Poster Session D: Non-Organ Specific: Economics, Public Policy, Allocation, Ethics
Session Type: Poster Session
Date: Tuesday, May 2, 2017
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:00pm-7:00pm
Presentation Time: 6:00pm-7:00pm
Location: Hall D1
Introduction:
With the implementation of the new Kidney Allocation System (KAS), deceased donor transplants from blood group A2 donors can now be implanted into blood group B recipients. However, the safety of this depends on low anti-A2 titer levels in the recipient. There is a lack of data as to the stability of these titers over time and therefore proper frequency of testing is unknown. This has limited the broad implementation of A2 to B transplants.
Methods:
From a single center's B waitlist and previous 2-year transplant record, blood group B patients were retrospectively identified who had at least two anti-A2 titer measurements. Anti-A2 titer values were collected along with information from the medical record related to possible sensitizing events. In order to account for the inherent imprecision of testing, a significant change in titer levels was defined as > 4-fold difference between measurements.
Results:
The sample included 48 adults (58% male; age 56 + 11 years). Average time between measurements was 13.31 + 6 months. Using the aforementioned criteria 35% (n=17) of patients had significant titer changes. Six patients had a significant decrease in titers, one of which was began immunosuppression and another received a blood transfusion between titers. The remaining 11 patients had a significant increase in titer levels. Between titers 4 underwent systemic antibiotic therapy (a marker of bacteremia) and 2 began hemodialysis, but the remaining 8 had no identifiable sensitizing events. Of the 31 patients with multiple titers that did not change significantly, 2 received a blood transfusion, 1 became pregnant, and 2 were treated with systemic antibiotics between measurements.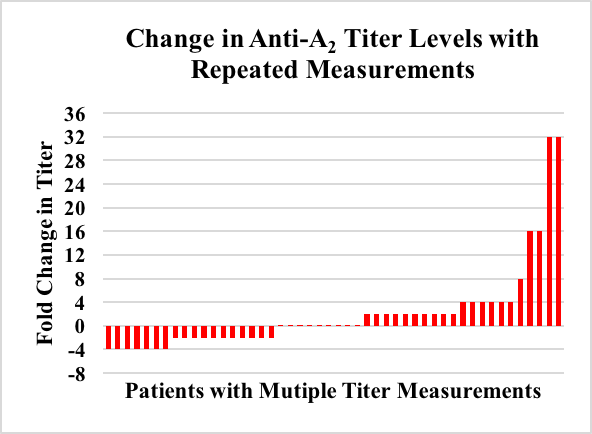 Conclusion:
Conclusion:
Over one third of anti-A2 titers show significant variability within 1 year of measurement. Careful medical history taking for hypothesized sensitizing events is not adequate to predict significant changes in titer levels. Frequent testing of anti-A2 titers is required to maintain adequate safety for patients listed for A2 to B transplants.
CITATION INFORMATION: Radomski S, Rosen-Bronson S, Diedrich B, Langeberg A, Li D, Awwad M, Grafals M, Cooper M, Gilbert A. It's Never too Early to Test: Frequent Anti-A2 Titer Monitoring in Blood Group B Patients is Necessary. Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Radomski S, Rosen-Bronson S, Diedrich B, Langeberg A, Li D, Awwad M, Grafals M, Cooper M, Gilbert A. It's Never too Early to Test: Frequent Anti-A2 Titer Monitoring in Blood Group B Patients is Necessary. [abstract]. Am J Transplant. 2017; 17 (suppl 3). https://atcmeetingabstracts.com/abstract/its-never-too-early-to-test-frequent-anti-a2-titer-monitoring-in-blood-group-b-patients-is-necessary/. Accessed May 6, 2026.« Back to 2017 American Transplant Congress
