Diagnosis and Prevalence of Strongyloidiasis in Solid Organ Transplant Patients
1Division of Infectious Diseases, Baylor College of Medicine, Houston, TX
2National School of Tropical Medicine, Baylor College of Medicine, Houston, TX.
Meeting: 2018 American Transplant Congress
Abstract number: C349
Keywords: Immunosuppression, Infection, Screening
Session Information
Session Name: Poster Session C: Transplant Infectious Diseases
Session Type: Poster Session
Date: Monday, June 4, 2018
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:00pm-7:00pm
Presentation Time: 6:00pm-7:00pm
Location: Hall 4EF
Background: Immunosuppression of a host with chronic Strongyloides stercoralis infection can lead to dissemination, with a fatality rate of up to 85%. Screening for S. stercoralis in transplant patients is done with commercial test (CT) crude-antigen enzyme-linked immunoassays (ELISAs). In comparison, NIE is a highly specific recombinant S. stercoralis-specific antigen developed and optimized at the National Institutes of Health. We used both assays to assess the prevalence of strongyloidiasis in transplant patients.
Methods: We prospectively recruited 150 patients from endemic regions who were pre- or post-solid organ transplant. Patients were consented, metadata gathered, and serum was sent for CT Strongyloides IgG ELISA. A recombinant NIE Ss IgG ELISA was performed on all serum samples at our institution.
Results: Preliminary data yielded a median age of 58 years. 79.1% of patients were from Latin America, 15.7% from Asia, and 5.2% from Africa. There were 59.7% pre-transplant patients and 40.3% post-transplant patients. 12.3% had a positive CT Strongyloides ELISA (85.7% pre-transplant, 14.3% post-transplant). NIE Ss IgG ELISA was positive in 3.8% of patients, (66.7% pre-transplant, 33.3% post-transplant). There was a significant increase in Ss IgG using the NIE Ss ELISA (geomean 19.27 vs 6.00 vs 4.32, p=0.0018). 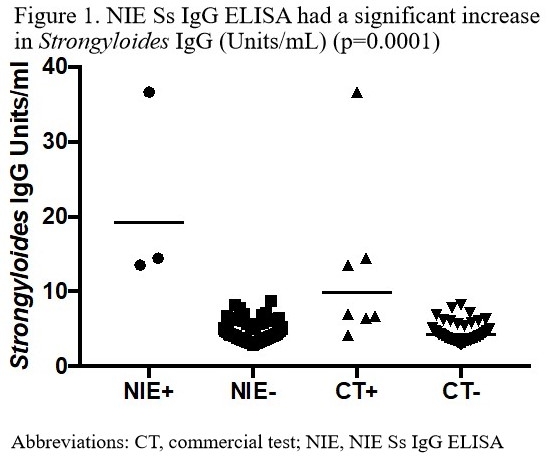 Eosinophilia was higher for NIE, CT ELISAs versus negative assays (geomean 575, 430, 131 cells/[micro]l respectively, p=0.0046). NIE Ss IgG ELISA had a 100% positive predictive value and a 92.6% negative predictive value (p=0.0012) for strongyloidiasis in this population.
Eosinophilia was higher for NIE, CT ELISAs versus negative assays (geomean 575, 430, 131 cells/[micro]l respectively, p=0.0046). NIE Ss IgG ELISA had a 100% positive predictive value and a 92.6% negative predictive value (p=0.0012) for strongyloidiasis in this population.
Conclusions: We found an endemic prevalence of strongyloidiasis at our institution in a transplant population. The NIE Ss IgG ELISA is specific for S. stercoralis and can decrease the false positive rate compared to commercial assays which rely on crude antigen from homogenized Strongyloides larvae that cross-reacts with other helminths. We recommend screening all at-risk solid organ transplant patients for strongyloidiasis, especially those with eosinophilia.
CITATION INFORMATION: Pritchard H., Hemmige V., Mejia R., Restrepo A. Diagnosis and Prevalence of Strongyloidiasis in Solid Organ Transplant Patients Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Pritchard H, Hemmige V, Mejia R, Restrepo A. Diagnosis and Prevalence of Strongyloidiasis in Solid Organ Transplant Patients [abstract]. https://atcmeetingabstracts.com/abstract/diagnosis-and-prevalence-of-strongyloidiasis-in-solid-organ-transplant-patients/. Accessed May 15, 2026.« Back to 2018 American Transplant Congress
