De Novo DSA with T Cell Mediated Rejection (TCMR) Leads to Early Allograft Scarring and Dysfunction.
UPMC, Pittsburgh
Meeting: 2017 American Transplant Congress
Abstract number: 181
Keywords: Alloantibodies, Graft function, Rejection
Session Information
Session Name: Concurrent Session: Pathways of Clinical Rejection
Session Type: Concurrent Session
Date: Sunday, April 30, 2017
Session Time: 4:30pm-6:00pm
 Presentation Time: 4:42pm-4:54pm
Presentation Time: 4:42pm-4:54pm
Location: E351
Detection of de novo DSA is associated with adverse outcomes in kidney transplant recipients (KTRs). The relationship between de novo DSA and subsequent TCMR and its impact on allograft survival is unknown. In this prospective longitudinal study, post-transplant DSA was compared to the development of TCMR and subsequent outcomes.
De novo DSA: DSA were analysed at 0,1,3,6,9,12 & 18mo. KTRs had protocol Bx at 3 & 12mo in addition to for-cause Bx. 79 (21%) of 372 KTRs transplanted between 1/13&12/14, developed de novo DSA (IS: Thymo, MPA+TAC & rapid steroid withdrawal). In 58/79 (73%) of these KTRs, DSA developed early(≤3m). 51% of KTRs who developed DSA had acute rejection versus 29% of KTRs without DSA (OR 2.54, p=0.002). Of the KTRs with DSA and rejection, 86% had DSA at or before the diagnosis of acute rejection while 14% developed DSA after acute rejection episode. Only 5 KTRs (12.5% vs. 87.5% TCMR) developed AMR and were excluded.
Renal Outcome: KTRs with DSA had significantly worse renal function at 12, 18 & 24m than those without DSA. KTRs with DSA+TCMR had worse Scr than those with either DSA/TCMR alone. Moreover, KTRs with DSA had significantly worse chronicity (IFTA & IF+i) scores than those without DSA. KTRs with DSA+TCMR had the worst chronicity scores than those with either DSA/TCMR alone (Fig 1). Thus, combination of DSA+TCMR is associated with poor allograft function and histology. Early diagnosis and treatment could improve outcomes.
Risk variables: KTRs with DSA+TCMR were younger (43 vs.55yrs, p=0.02) and had a higher incidence of DGF (35%vs.14.8%, p=0.001) when compared to those with DSA alone and both were independent risk factors (recipient age OR 0.95, p<0.05; DGF OR 6.9, p=0.02). Cumulative MFI for the DSA were significantly higher in the DSA+TCMR sub-group (7106 vs.3212, p=0.03).
Conclusions:
i) de novo DSA with TCMR is associated with worse renal function and chronic allograft injury than DSA alone, suggesting that DSA is a marker for a heightened cellular alloresponse and early detection may preserve graft function.
ii) In KTRs with de novo DSA, variables such as young age and DGF are important risk factors for the occurrence of TCMR.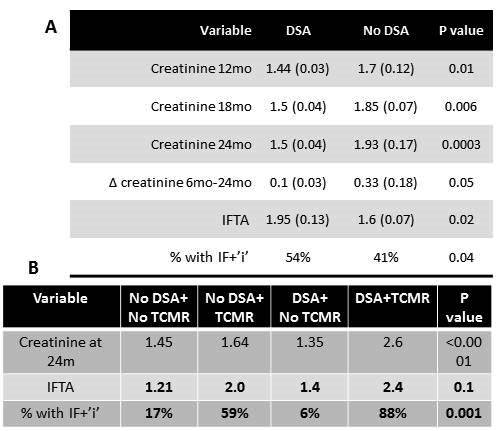
CITATION INFORMATION: Cherukuri A, Sharma A, Mangiola M, Sood P, Randhawa P, Zeevi A, Mehta R, Hariharan S. De Novo DSA with T Cell Mediated Rejection (TCMR) Leads to Early Allograft Scarring and Dysfunction. Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Cherukuri A, Sharma A, Mangiola M, Sood P, Randhawa P, Zeevi A, Mehta R, Hariharan S. De Novo DSA with T Cell Mediated Rejection (TCMR) Leads to Early Allograft Scarring and Dysfunction. [abstract]. Am J Transplant. 2017; 17 (suppl 3). https://atcmeetingabstracts.com/abstract/de-novo-dsa-with-t-cell-mediated-rejection-tcmr-leads-to-early-allograft-scarring-and-dysfunction/. Accessed June 30, 2025.« Back to 2017 American Transplant Congress
