Critical Role of Notch-2 Signaling in Alloantibody Production
Transplantation Research Center, Brigham &
Women's Hospital, Harvard Medical School, Boston
Genentech, South San Francisco
Meeting: 2013 American Transplant Congress
Abstract number: 470
The role of B cells in the alloimmune response is increasingly recognized, while alloantibody production remains a major barrier to the improvement of long-term graft survival. We have previously shown that Notch signaling plays a crucial role in peripheral T cell differentiation; however, little is known about the importance of Notch in B cell differentiation and plasma cell generation in transplantation. We sought to investigate the role of Notch-2 signaling therein using a novel, selective antibody against the Notch-2 receptor (aNotch-2; Wu et al, Nature 2010) in a stringent, fully MHC mismatched cardiac transplant model (Balb/c⇒B6).
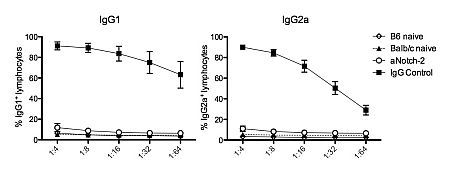
B6 recipients treated with aNotch-2 for 6 days demonstrated significant, albeit modest, prolongation of graft survival compared to IgG-treated controls (MST 10 vs 7 days; p=0.0023). Strikingly, treatment with aNotch-2 effected a 10-fold reduction in marginal zone B cells and a 3-fold reduction in transitional zone type 2 (T2) B cells compared to IgG-treated controls (p<0.0001 and p=0.0079, respectively), while follicular B cells were increased (78.7±1.3 vs 64.4±0.2% in controls; p=0.0002). Furthermore, there was a significant reduction in the population of plasma cells (B220–CD138+) in both the spleen and bone marrow of aNotch-2-treated mice (0.46±0.03 and 0.2±0.004 vs 1.7±0.3 and 0.3±0.01%; p=0.047 and p=0.01, respectively). Most remarkable, however, was the profound inhibition of alloantibody production seen in mice treated with aNotch-2 (Figure; IgG1 and IgG2a).
Blockade of Notch-2 was also shown to favorably affect the cytokine milieu: Luminex analysis of supernatants collected at 48hrs revealed reduced levels of inflammatory cytokines, including IFN-Γ, IL-6, IL-17, IL-1Β and TNF-Α, as compared to IgG-treated controls.
These data reveal a crucial role of Notch-2 in B cell differentiation and function in transplantation, particularly in the production of alloantibodies, and indicate that selective blockade of Notch-2 may be a useful therapeutic strategy to prevent the antibody-mediated injury so resistant to current therapies.
Siebel, C.: Employee, Genentech, Inc.
To cite this abstract in AMA style:
Magee C, Shimizu T, Ohori S, Siebel C, Najafian N, Riella L. Critical Role of Notch-2 Signaling in Alloantibody Production [abstract]. Am J Transplant. 2013; 13 (suppl 5). https://atcmeetingabstracts.com/abstract/critical-role-of-notch-2-signaling-in-alloantibody-production/. Accessed May 25, 2026.« Back to 2013 American Transplant Congress
