Co-Stimulation Blockade to Minimize and Withdraw Long Term Calcineurin Inhibitors (CNI) in Simultaneous Pancreas and Kidney Transplant (SPK).
1UCSF, San Francisco
2UAB, Birmingham, AL
3Rho Inc, Chapel Hill, NC
4NIH/NIAID, Bethesda, MD
5UW, Madison, WI
6IU, Indianapolis, IN
7Emory University, Atlanta, GA
Meeting: 2017 American Transplant Congress
Abstract number: 566
Keywords: Immunosuppression, Infection, Pancreas transplantation, Rejection
Session Information
Session Name: Concurrent Session: Late Breaking
Session Type: Concurrent Session
Date: Tuesday, May 2, 2017
Session Time: 4:30pm-6:00pm
 Presentation Time: 4:30pm-4:42pm
Presentation Time: 4:30pm-4:42pm
Location: E353C
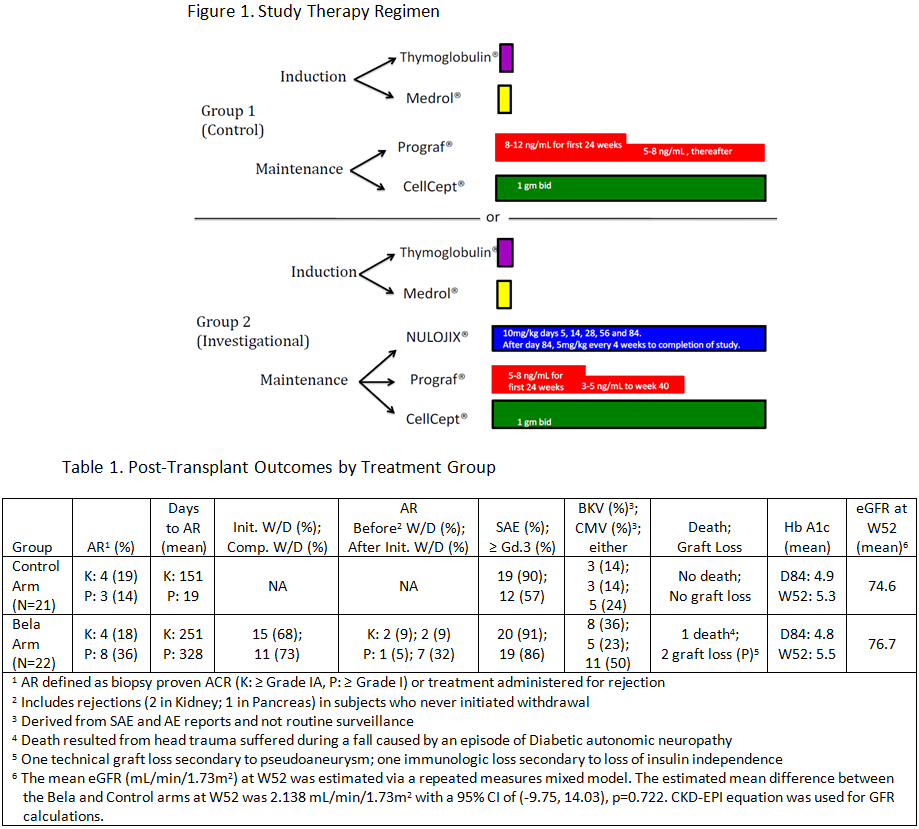
The NIH multicenter CTOT-15 trial tested the hypothesis that use of belatacept (bela) could minimize or eliminate long-term CNI use in simultaneous pancreas and kidney transplant (SPK) recipients.
Methods: Primary SPK transplant (tx) recipients were randomized to 2 groups (Figure 1). Subjects in the in the Bela arm were eligible for CNI withdrawal at week 40 post tx if they had stable pancreatic and renal function over the preceding 90 days, had no evidence of cellular or antibody-mediated rejection in the first 280 days, and had no DSA at week 36. Selected endpoints at 52 weeks or 60 days post tacrolimus withdrawal included eGFR (primary), the incidence and severity of rejection, Hgb A1C, rates of insulin independence, and safety measures.
Results: The number of patients enrolled in each arm and selected outcome measures are listed in Table 1. 15/22 of subjects enrolled in Bela arm were eligible and underwent attempted CNI withdrawal. SAEs were common in both arms. There was one pancreas allograft lost in the Bela arm due to bleeding from an arterial pseudoaneurysm resulting in pancreatectomy and one lost due to allograft rejection and return to insulin dependence with stable renal allograft function. Renal function was equivalent in both groups. The high rate of pancreas AR following CNI withdrawal prompted the DSMB to stop CNI withdrawal.
Conclusions: SAE rates for both arms were equally high, consistent with challenges of SPK. AR rates for both arms for both organs were low while maintained on either standard or low-dose CNI + bela. However, the high rates of pancreas AR in the bela arm following withdrawal of CNI demonstrate the importance of CNI in SPK tx. Although low-dose CNI combined with belatacept provides effective immunosuppression in SPK, the high incidence of infections may outweigh the potential long-term benefits of CNI minimization.
CITATION INFORMATION: Stock P, Mannon R, Armstrong B, Ikle D, Robian M, Morrison Y, Odorico J, Fridell J, Mehta A, Newell K. Co-Stimulation Blockade to Minimize and Withdraw Long Term Calcineurin Inhibitors (CNI) in Simultaneous Pancreas and Kidney Transplant (SPK). Am J Transplant. 2017;17 (suppl 3).
To cite this abstract in AMA style:
Stock P, Mannon R, Armstrong B, Ikle D, Robian M, Morrison Y, Odorico J, Fridell J, Mehta A, Newell K. Co-Stimulation Blockade to Minimize and Withdraw Long Term Calcineurin Inhibitors (CNI) in Simultaneous Pancreas and Kidney Transplant (SPK). [abstract]. Am J Transplant. 2017; 17 (suppl 3). https://atcmeetingabstracts.com/abstract/co-stimulation-blockade-to-minimize-and-withdraw-long-term-calcineurin-inhibitors-cni-in-simultaneous-pancreas-and-kidney-transplant-spk/. Accessed June 3, 2026.« Back to 2017 American Transplant Congress