Role of Increased Autophagic Flux in Renal Tubular Epithelial Cells Undergoing Warm Reperfusion (WR) after Cold Ischemia (CI), The
UC Denver, Aurora, CO
Meeting: 2013 American Transplant Congress
Abstract number: D1668
Delayed graft function (DGF) is primarily caused by cold ischemia (CI) and warm reperfusion (WR). The mechanism by which CI/WR causes renal tubular epithelial cell (RTEC) death is not known. Autophagy is a cell-survival strategy used by cells during stresses such as ischemia and reperfusion. The relationship between apoptosis and autophagy during CI/WR of donor kidneys is unknown. We hypothesized that increased autophagic flux and apoptosis occur during CI/WR of RTE cells. Methods: RTECs (LLC-PK1) were subjected to CI in UW at 4°C for 24h. To simulate rewarming, UW was replaced with DMEM containing bovine serum at 37°C for 24h. LLCPK cells incubated at 37°C served as controls. Immunoblot and densitometry were used to assess active caspase-3 (17kDa), caspase-1 (45kDa) and LC3–II (14kDa), a marker of autopahgic flux. Autophagic flux is measured by comparing LC3-II protein expression with and without Bafilomycin, a lysosomal inhibitor, and wortmannin, a PI3 kinase inhibitor. Cells were incubated with Bafilomycin (150nM) and wortmanin (250nM) 1 hour before CI. Results: During CI, caspase-1 was not increased whereas apoptosis and active caspase 3 were significantly increased vs. controls (Table 1).
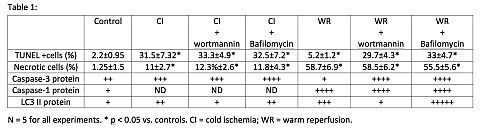
Treatment with Bafilomycin and wortmannin did not change LC-3 II protein expression during CI indicating that autophagic flux was not increased. In contrast, during WR, apoptosis and caspase-3 did not increase whereas caspase-1 and necrosis increased significantly. Treatment with Bafilomycin during WR resulted in significantly more LC-3II expression (indicating increased autophagic flux)and apoptosis but had no effect on necrosis or caspase-1. Conclusions: During CI, caspase-3 and apoptosis are increased but autophagic flux is not. Bafilomycin and wortmannin had no effect on apoptosis or necrosis during CI. In contrast, autophagic flux, caspase-1 and necrosis are increased during WR. Bafilomycin and wortmannin increased caspase-3 and apoptosis during WR, but had no effect on caspase-1 and necrosis. These results suggest autophagic flux protects from apoptosis during WR but does not prevent caspase-1 activation nor necrotic cell death. Autophagy inhibitors should be used with caution during donor kidney reperfusion.
To cite this abstract in AMA style:
Jani A, Jain S, Nydam T, Edelstein C. Role of Increased Autophagic Flux in Renal Tubular Epithelial Cells Undergoing Warm Reperfusion (WR) after Cold Ischemia (CI), The [abstract]. Am J Transplant. 2013; 13 (suppl 5). https://atcmeetingabstracts.com/abstract/role-of-increased-autophagic-flux-in-renal-tubular-epithelial-cells-undergoing-warm-reperfusion-wr-after-cold-ischemia-ci-the/. Accessed July 5, 2025.« Back to 2013 American Transplant Congress
