Effects of Various Therapeutic Levels of Tacrolimus on Early Post Transplant Outcomes
University of Florida, Gainesville
Meeting: 2013 American Transplant Congress
Abstract number: B941
Background: In striving to achieve early tacrolimus therapeutic levels, there is a risk of overshooting and achieving supratherapeutic or toxic levels. We studied our patients to determine the detrimental effects of supratherapeutic levels on early post-transplant outcomes.
Methods:We included all adult solitary 1st kidney transplants at our center between January 2004 and May 2012. Live donor, donation after cardiac death, and expanded criteria donor kidney transplants were excluded. Patients were divided into 3 groups: those with peak post-transplant tacrolimus levels between 10-12 ng/mL (deemed therapeutic at our institution), 12-15 ng/mL and >15 ng/mL in the first week post-transplant. Patients with subtherapeutic (<10 ng/mL) levels were excluded. Outcomes were compared across the three groups and multivariate regression analysis was applied. Primary studied outcomes were incidence of delayed graft function (DGF), defined as need for dialysis in the first week post-transplant. Secondary outcomes included length of stay (LoS) post-transplant, estimated glomerular filtration rate (eGFR) by the MDRD formula at end of first week, hyperglycemia in first month, and acute rejection (AR) at 1- and 3-months post-transplant. Adjusted covariates included: donor’s age, race, gender, and recipient’s age, gender, race, BMI, end stage renal disease, dialysis length, cold ischemia time, HLA mismatch, and induction medications.
Results: There were no significant differences in baseline characteristics including use of antithymocyte globulin (ATG). Important results are shown in the table:
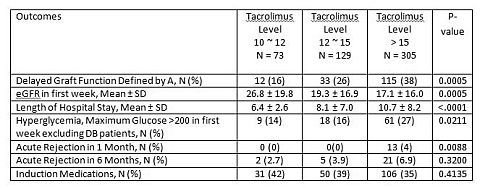
Above results remained significant even after adjusting for other potential risk factors. Multivariate regression analysis showed that the 12-15 ng/mL group was significantly different from the 10-12 ng/mL group in LoS alone, all other outcomes were nonsignificant.
Conclusions: In our analysis, a supratherapeutic tacrolimus level of >15 ng/mL was significantly associated with impaired renal function and its associated complications. Further study is warranted to determine the safest strategy to accelerate early immunosuppression dosing to ptimally balance toxicity with adequate graft protection.
To cite this abstract in AMA style:
Rehman S, Wen X, Casey M, Santos A, Andreoni K. Effects of Various Therapeutic Levels of Tacrolimus on Early Post Transplant Outcomes [abstract]. Am J Transplant. 2013; 13 (suppl 5). https://atcmeetingabstracts.com/abstract/effects-of-various-therapeutic-levels-of-tacrolimus-on-early-post-transplant-outcomes/. Accessed July 18, 2025.« Back to 2013 American Transplant Congress
