Quantitative Hepatitis C Virus (HCV) RNA in Plasma and Kidney Tissue in HCV Antibody Positive Donors.
1Surgery, Transplant, Hershey Medical Center, Hershey, PA
2Pathology, Hershey Medical Center, Hershey, PA
3OPO, Gift of Life Donor Program, Philadelphia, PA.
Meeting: 2016 American Transplant Congress
Abstract number: B196
Keywords: Cadaveric organs, Hepatitis C, Kidney, Risk factors
Session Information
Session Name: Poster Session B: Kidney Transplantation: KDPI, HCV/Matching, Donor Age
Session Type: Poster Session
Date: Sunday, June 12, 2016
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:00pm-7:00pm
Presentation Time: 6:00pm-7:00pm
Location: Halls C&D
Background: Currently HCV antibody positive deceased donors renal allografts are not considered for transplantation in HCV negative recipients. With recent development of anti-HCV therapy, utilization of such allografts may be considered if the viral load in the renal allograft is known to be negative or minimal.
Aim: To examine the viral load in renal allograft tissue after routine flush with cold UW solution and compare with plasma HCV RNA from the same donor.
Material and Method: Plasma and 28 kidney samples were collected from 14 HCV antibody positive donors after routine cold flush. Quantitative RT-PCR: COBAS AMPLICOR automated analyzer for plasma and TaqMan assay for kidney tissue (Engle RE, et al 2008). For kidney HCV RNA quantitation, approximately 50-100 mg kidney tissue was used to extract RNA by TRIzol. After reverse transcription, cDNA from 100 ng cellular RNA equivalent was used for PCR. Cycle threshold (CT) of tissue samples were compared to the CT generated from the plasma samples with known HCV RNA levels. HCV international unit (IU) was normalized per 50 nL for both plasma and kidney, by dividing plasma IU/mL by 20,000 and considering that 1, 50 mg kidney tissue typically contains 100 ng RNA, and 2) 50 mg tissue is equivalent to 50nL volume (1g tissue is approximately 1mL).
Results:
1) 14 Donors who were HCV antibody positive, 11 (78.6%) donor's serum were positive for HCV RNA. 2) Out of 28 kidneys from HCV antibody positive donor; 16 (57.1%) were positive for HCV RNA. 3) Mean (± SD) HCV RNA in plasma was 152±284 IU/50 nL plasma and 1425 IU/50 nL kidney tissue.
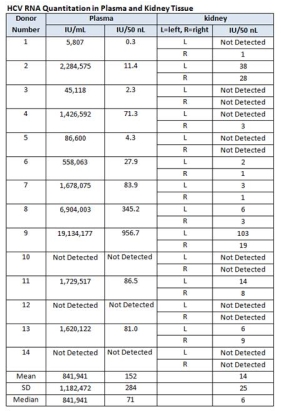
Conclusion: 79 % of serum and 57% of renal grafts are positive for HCV RNA, from HCV antibody positive donor. The mean quantitative HCV was approximately 10-fold lower in kidney tissue than in plasma. The risk may be further reduced with multiple flushes. Future studies are required to improve utilization of HCV positive donors with newly developed anti HCV therapy.
CITATION INFORMATION: Shike H, Kadry Z, Shah R, Imamura Y, Greene W, Gaspari J, Riely T, Nathan H, Hasz R, Jain A. Quantitative Hepatitis C Virus (HCV) RNA in Plasma and Kidney Tissue in HCV Antibody Positive Donors. Am J Transplant. 2016;16 (suppl 3).
To cite this abstract in AMA style:
Shike H, Kadry Z, Shah R, Imamura Y, Greene W, Gaspari J, Riely T, Nathan H, Hasz R, Jain A. Quantitative Hepatitis C Virus (HCV) RNA in Plasma and Kidney Tissue in HCV Antibody Positive Donors. [abstract]. Am J Transplant. 2016; 16 (suppl 3). https://atcmeetingabstracts.com/abstract/quantitative-hepatitis-c-virus-hcv-rna-in-plasma-and-kidney-tissue-in-hcv-antibody-positive-donors/. Accessed April 23, 2024.« Back to 2016 American Transplant Congress
