Introducing a Mobile Phone Application to Transplant Patients to Encourage Medication Compliance and Education in This At-Risk Cohort.
A. Zanetti Yabur, A. Rizzo, J. Graham, J. Rocca.
Abdominal Transplant Surgery, Montefiore Medical Center, Bronx, NY.
Meeting: 2016 American Transplant Congress
Abstract number: D281
Keywords: Kidney/liver transplantation, Patient education
Session Information
Session Name: Poster Session D: Psychosocial and Treatment Adherence
Session Type: Poster Session
Date: Tuesday, June 14, 2016
Session Time: 6:00pm-7:00pm
 Presentation Time: 6:00pm-7:00pm
Presentation Time: 6:00pm-7:00pm
Location: Halls C&D
Introduction: The efficacy of a mobile phone application in promoting medication adherence and education was investigated to evaluate its utility in assisting liver and kidney transplant patients in managing immunosuppression.
Methods: Patients completed 3 surveys peri-operatively; a Beliefs about Medicine Questionnaire (BMQ) was used to establish baseline predilections about compliance in the early post-operative period. Patients were also assessed using the Morisky Medication Adherence Scale (MMAS-8) and tested on their medication recall by writing down their immunosuppressive medications approximately 3-months post-operatively. Serum tacrolimus levels and rejection episodes were also denoted as a surrogate for medication adherence.
Results: Overall, our transplant patients had a negative view of medications (BMQ score > 47 = negative beliefs about medicine); BMQ scores between app users (n=21) and non-users (n=53); (52.4 v. 58.29; p = 0.09). Also, there was no difference in mean tacrolimus levels between app users and non-users; (9.5 ug/ml v. 9.52 ug/ml; p=NS). However, app users had a higher educational component score than app non users (2.66 v. 1.87; p= 0.005). Interestingly, app users demonstrated a tendency toward poorer adherence behavior; MMAS-8 (1.33 v. 0.5; p=0.0027). The application users had one rejection episode, while the non-users had three rejection episodes (p=NS). 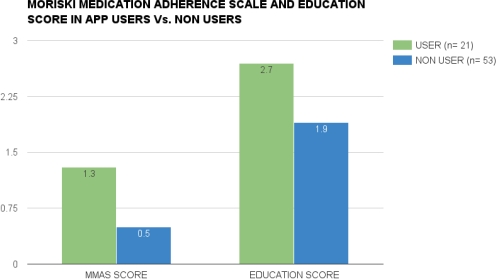
Conclusion: Admittedly, the introduction of an app to assist in medication compliance requires further investigation, longer study periods, and a larger volume of patients to determine its usefulness in this cohort. However, in this pilot study app users seemed to gain an educational benefit as it relates to their ability to accurately document their immunosuppressive treatment. Moreover, perhaps app users gained the most benefit as they were more likely to have a non-compliant behavior in the context of a negative belief about their medication in their peri-operatively period.
CITATION INFORMATION: Zanetti Yabur A, Rizzo A, Graham J, Rocca J. Introducing a Mobile Phone Application to Transplant Patients to Encourage Medication Compliance and Education in This At-Risk Cohort. Am J Transplant. 2016;16 (suppl 3).
To cite this abstract in AMA style:
Yabur AZanetti, Rizzo A, Graham J, Rocca J. Introducing a Mobile Phone Application to Transplant Patients to Encourage Medication Compliance and Education in This At-Risk Cohort. [abstract]. Am J Transplant. 2016; 16 (suppl 3). https://atcmeetingabstracts.com/abstract/introducing-a-mobile-phone-application-to-transplant-patients-to-encourage-medication-compliance-and-education-in-this-at-risk-cohort/. Accessed April 17, 2024.« Back to 2016 American Transplant Congress
