Informing Informed Consent for High KDPI Kidneys.
A. Nishio Lucar, D. Keith, G. Vranic.
Medicine, University of Virginia, Charlottesville, VA.
Meeting: 2016 American Transplant Congress
Abstract number: 559
Keywords: Allocation, Informed consent, Kidney transplantation, Outcome
Session Information
Session Name: Concurrent Session: The High KDPI Kidney: Outcomes and Optimal Utilization
Session Type: Concurrent Session
Date: Tuesday, June 14, 2016
Session Time: 4:30pm-6:00pm
 Presentation Time: 4:30pm-4:42pm
Presentation Time: 4:30pm-4:42pm
Location: Ballroom C
The standard approach to informed consent for high KDPI (KDPI 86-100%) deceased donor kidneys (DDK) relies on anticipated graft half-life and may not provide candidates with accurate assessment of potential outcomes.
We evaluated 6 month outcomes among DDK recipients from different donor allocation groups: ideal (KDPI 0-20%), standard (KDPI 21-85%) and high KDPI kidneys. We correlated these with long-term patient survival. All adult DDK recipients between January 1, 2005 and December 31, 2012 were identified in the SRTR database. KDPI was calculated using 2013 as a reference. We determined 6-month eGFR by CKD-EPI and death censored graft loss by KDPI group. We considered a suboptimal outcome any eGFR <30 ml/min/1.72m2 or failed-graft not due to recipient death at 6 months. Death with graft function was not deemed a suboptimal outcome.
We analyzed 78,022 DDK recipients after excluding 383 recipients lost to follow-up and 2.9% lacking a 6-month creatinine. Graft outcome by KDPI at 6-month is shown in Table 1.
| KDPI group | Optimal Outcome | CKD stage 4 or 5 | Death Censored Graft Loss |
| Ideal Kidney (%) | 96.2 | 1.9 | 1.9 |
| Standard Kidney (%) | 89.7 | 6.8 | 3.6 |
| High KDPI Kidney (%) | 74.7 | 18.2 | 7.1 |
25.3% of high KDPI kidneys recipients had suboptimal outcome versus 10.4% of standard and 3.8% of ideal kidneys. Figure 1 shows the distribution of recipient CKD stage by KDPI group.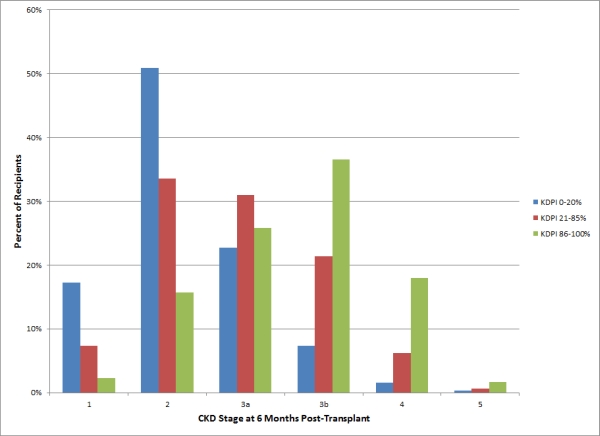 Recipients of a high KDPI kidney who had CKD stage 4 or 5 or death censored graft loss at 6 months had reduced patient survival by about 15% and 28% respectively.
Recipients of a high KDPI kidney who had CKD stage 4 or 5 or death censored graft loss at 6 months had reduced patient survival by about 15% and 28% respectively.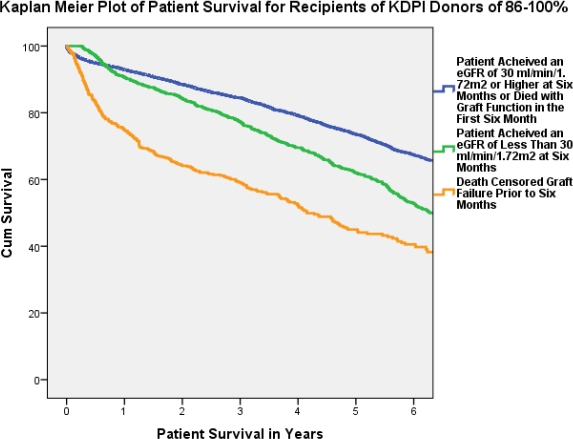
Most high KDPI DDK recipients did reasonably well but 25% of them had poorer outcomes. The effect is sustained and portends decreased patient survival. Candidates considering high KDPI organs should be better informed of the risks of accepting these organs versus the potential benefit of shortening waiting time.
CITATION INFORMATION: Nishio Lucar A, Keith D, Vranic G. Informing Informed Consent for High KDPI Kidneys. Am J Transplant. 2016;16 (suppl 3).
To cite this abstract in AMA style:
Lucar ANishio, Keith D, Vranic G. Informing Informed Consent for High KDPI Kidneys. [abstract]. Am J Transplant. 2016; 16 (suppl 3). https://atcmeetingabstracts.com/abstract/informing-informed-consent-for-high-kdpi-kidneys/. Accessed April 26, 2024.« Back to 2016 American Transplant Congress
